
Gemeinsam mit CELEST war das IAM-ESS im Februar 2026 erfolgreich im Wettbewerb des Landes Baden-Württemberg um Fördermittel aus dem Europäischen Fonds für regionale Entwicklung (EFRE). Mit CELEST-CERL (CELEST Circular Economy Recycling Lab) entsteht am KIT eine Forschungsinfrastruktur zur Entwicklung nachhaltiger Recyclingstrategien für kritische Materialien, insbesondere Altbatterien. CERL schafft die Grundlage für die Entwicklung neuartiger Recyclingverfahren. Im Fokus stehen ein mechanochemisches und ein halothermisch genanntes Verfahren zur Rückgewinnung wertvoller Rohstoffe bei moderaten Bedingungen. Langfristig sollen diese Methoden auch auf weitere Rezyklate ausgeweitet werden.
Link
Forschende des KIT am IAM-ESS haben ein neuartiges Verfahren zum Recycling von Altbatterien entwickelt: Halometallurgie. Obwohl der Prozess auf kostengünstigen und weit verbreiteten Salzen wie Natriumchlorid und Kaliumchlorid basiert, liegt ihm ein faszinierendes wissenschaftliches Prinzip zugrunde, das die Wechselwirkung mit der Batterie-Schwarzmasse bestimmt. Aufgrund seiner Einfachheit und inhärenten Sicherheit könnte die Halometallurgie zur Dezentralisierung des Recyclingmarktes beitragen. Weitere Informationen finden sich in der zugehörigen Publikation in Chemistry of Materials, die als ACS Editor’s Choice ausgewählt wurde.
Publikation:
In der Initiative BRIDGE untersuchen Forschende des Karlsruher Instituts für Technologie (KIT) und der Bundesanstalt für Geowissenschaften und Rohstoffe (BGR) gemeinsam mit chilenischen Partnern entsprechende Lagerstätten in der Atacama-Wüste. Ziel ist es, ihr Rohstoffpotenzial zu analysieren und Strategien für einen schonenden Abbau zu entwickeln. Das IAM-ESS prüft beispielsweise Verfahren der Direktextraktion, bei denen kritische Rohstoffe mithilfe selektiver Materialien oder chemischer Prozesse unmittelbar aus den Lösungen abgeschieden werden.
KIT-Presseinformation
Gemeinsam mit CELEST war das IAM-ESS im Februar 2026 erfolgreich im Wettbewerb des Landes Baden-Württemberg um Fördermittel aus dem Europäischen Fonds für regionale Entwicklung (EFRE). Mit CELEST-CERL (CELEST Circular Economy Recycling Lab) entsteht am KIT eine Forschungsinfrastruktur zur Entwicklung nachhaltiger Recyclingstrategien für kritische Materialien, insbesondere Altbatterien (Gesamtbudget: 1.53Mio EUR). CERL schafft somit die Grundlage für die Entwicklung neuartiger Recyclingverfahren. Im Fokus stehen ein mechanochemisches und ein halothermisch genanntes Verfahren zur Rückgewinnung wertvoller Rohstoffe bei moderaten Bedingungen. Langfristig sollen die Methoden auch auf weitere Rezyklate ausgeweitet werden, denn für innovative Batteriekonzepte mit geringem Materialwert sind klassische Methoden oft nicht nachhaltig genug. Im Rahmen der EFRE-Ausschreibung war das KIT mit drei weiteren Vorhaben erfolgreich.
Link
Im Rahmen des CuLiWell: Stakeholderworkshop haben sich Forschende und Vertreterinnen und Vertreter aus Politik, Industrie und Nichtregierungsorganisationen zur Ko-Extraktion von kritischen Rohstoffen und Wärme aus geothermischen Fluiden ausgetauscht. Das IAM-ESS hat seine Expertise in der Lithium-Extraktion eingebracht.

We would like to congratulate Noah Keim, doctoral student at IAM-ESS in the “Electrode Coating” working group, who has been selected for the Junior Experts Exchange Program, which this year focused on “Battery Research for Carbon Neutrality.” Together with five other experts, the Junior Experts were invited for a trip to Japan and given an in-depth insight into the latest research in both industry (visits to Panasonic, Horiba, BASC, and PPES (a joint venture between Toyota and Panasonic)) and academia (Kyoto University and Tokyo University.

Sylvio Indris receives the Distinguished Lecture award at the eScience Summit Forum at Nankai University in Tianjin, China, on October 10, 2025.
The title of his lecture was “Ion Dynamics in Li, Na, and Mg Solid Electrolytes as Observed with NMR Spectroscopy”

For the second time, KIT (IAM-ESS) is proud to have hosted the Symposium on Direct Lithium Extraction on 9 – 10 October 2025. The event, which was directed by the scientific spokesperson, Dr. Fabian Jeschull, brought together leading experts from research and industry. The symposium, which spanned two days, covered the latest advancements in DLE technology, ranging from pioneering research to practical applications, with a focus on lithium extraction from salar and unconventional brines. With approximately 100 participants from both academic and industry backgrounds, the event provided a valuable opportunity to enhance collaboration between research and practice in this rapidly evolving field. Great thanks to the organization team: Fabian Jeschull, Martina Gamba, and Isabelle Südmeyer as well as the advisory board, Dr. Valentin Goldberg (BWG Geochemische Beratung GmbH, Germany) and Dr. Jonas Scottmann (University of Oslo, Norway) and to Laura Herrmann and Joscha Silas Fürniß from EnBW for hosting us at the Bruchsal geothermal power plant and DLE pilot.

Felix Bauer (IAM-ESS), a doctoral student from the Carbon-Based Materials working group, was awarded a poster prize for his work “Deciphering the Effect of Surface Oxygen Functionalities on Hard Carbon” at the International Conference on Sodium Ion Batteries (ICNaB 2025) in Sydney.

Das Titelbild der aktuellen Ausgabe von Crystal Growth & Design zeigt einen neuartigen Siloxan-verbrückten, dianionischen linearen Co(II)₃ Komplex. Aufgrund der ferromagnetisch gekoppelten Co-Momente weist dieser Einzelmolekülmagnet eine vergleichsweise langsame magnetische Relaxation auf. Die experimentellen Ergebnisse und theoretischen Berechnungen dazu entstanden durch Mitwirken eines Wissenschaftlers vom IAM-ESS in Kooperation mit Wissenschaftlerinnen und Wissenschaftern vom Indian Institute of Technology Madras und der National University of Singapore und sind als Artikel in Crystal Growth & Design (Vol 25/Issue 17) veröffentlicht.
Link zu Artikel:
Prof. Dr. Siegbert Schmid receives the International Excellence Fellowship from the Karlsruhe Institute of Technology (KIT). Prof. Schmid is currently a professor at the University of Sydney and will be completing his stay at the Institute for Applied Materials – Energy Storage Systems (IAM-ESS) at KIT under the direction of Prof. Dr. Helmut Ehrenberg, who is also POLiS spokesperson and principal investigator.

Während des Upcell Alliance Events in Barcelona on June 27, Noah Keim received the “2025 Upcell Young Scientist Award” for his doctoral thesis. This award recognizes the best dissertation related to batteries. Noah Keim from IAM-ESS focused his work on the processing of environmentally friendly binders that can be processed in an aqueous medium.

Solid State Ionics conference in London (July 15-19, 2024):
Prof. John Kilner (President of the Solid State Ionics Society) gives the poster award certificate to Dr. Mareen Schaller for her poster “Ion dynamics of multicationic substituted High-Entropy Argyrodite Superionic Conductors”.
Link zum Poster
The annual POLiS Conclave took place from June 23 to 23, 2025 in the heart of Karlsruhe. Around 130 scientists from Karlsruhe Institute of Technology, Ulm University and Justus Liebig University Giessen as well as international guest scientists met for two days to discuss the latest research trends in battery research and get an insight into the second funding period of the Cluster.
The agenda featured a total of 5 keynote presentations, 25 regular talks and three poster sessions, in which a total of over 50 POLiS members presented their research findings. Also this year, a poster prize was awarded, voted for by the participants.

The joint battery research network POLiS (Post Lithium Storage) has prevailed in the competition and remains a cluster of excellence. POLiS scientists have been conducting research into innovative battery materials and storage technologies since 2018. In the upcoming funding period from 2026 to 2032, the researchers will focus on the realisation of full cells and the interactions between the battery components along the entire cell. In the "Post Lithium Storage" cluster of excellence at KIT, the University of Giessen and Ulm University, which was approved initially in 2018, scientists – among these Prof. Helmut Ehrenberg, Prof. Anass Benayad, Prof. Daria Mikhailova, Dr. Fabian Jeschull and Dr. Anna Smith from IAM-ESS - are researching pioneering solutions for electrochemical energy storage (EES). The aim is to develop lithium-free batteries that are safe, powerful and sustainable. Further cooperation partners are the Centre of Solar Energy and Hydrogen Research Baden-Württemberg (ZSW) and Helmholtz Zentrum Berlin (HZB).

Forscher vom IAM-ESS (Dr. Michael Knapp, Dr. Sylvio Indris, Dr. Valeriu Mereacre) hatten bei ihrem Forschungsaufenthalt für eine Messreihe im ALBA Synchrotron die Ehre, António Costa, den Präsidenten des Europäischen Rates, willkommen zu heißen. In Begleitung von Núria Montserrat, Consellera de Recerca i Universitats; Javier Selva, Generaldirektor für Wissenstransfer, und Caterina Biscari, Direktorin von ALBA, besichtigte Präsident Costa die Einrichtungen und informierte sich über unsere Mission: Talente ausbilden und fördern Nutzern weltweit hochwertige Dienstleistungen anbieten Spitzenforschung in ganz Europa vorantreiben. Der Besuch unterstreicht die entscheidende Rolle großer Forschungsinfrastrukturen wie ALBA für die Förderung wissenschaftlicher Exzellenz, Zusammenarbeit und Wettbewerbsfähigkeit in Europa.
Link
Dr. Daria Mikhailova has recently joined Karlsruhe Institute of Technology (KIT) as professor for “Materials for Sustainable Electronic Energy Storage” at the Institute for Applied material – Energy Storage Systems (IAM-ESS). During his career before his arrival to KIT, she was Group Leader at Max-Planck Institute for hemical Physics of Solids (MPI CPfS), Dresden / Germany and of the Electrochemical Energy Storage group at IFW Dresden. The focus of her research group is on electrode materials for sodium and magnesium batteries. Watch the video to find out more on her reseach.

Around 25 researchers from the POLiS Cluster of Excellence and the Graduate School Electrochemical Energy Storage (GS-EES) – including IAM-ESS colleagues - had the great opportunity to visit the Center for Solar Energy and Hydrogen Research (ZSW) in Ulm - a key player in applied battery research and technology transfer in Germany.

Since April 2025, Dr. Anass Benayad has been assigned as professor for ´interfacial processes´ at the Institute for Applied material – Energy Storage Systems (IAM-ESS). During his career before his arrival to KIT, he was senior researcher at Samsung Advanced Institute of technology (SAIT) from 2006 to 2013 in South Korea, then Director of research at the French Atomic Energy Commission in the department of innovation for renewable energies and nanomaterial (CEA-Liten) from 2013 to 2025. He was in charge of surface characterization center at the platform of nano-characterization (PFNC). His research focusses on the investigation of the behavior of ions and electrons at the various interfaces within battery materials developing operando photoemission spectroscopy.

The project “Sodium-Ion-Battery Germany Research – SIB:DE FORSCHUNG,” funded by the Federal Ministry of Education and Research (BMBF), aims to evaluate the suitability of sodium-ion battery technology (SIB) for the European energy and mobility transition and to achieve rapid industrial implementation. To this end, 21 national partners from science and industry are pooling their expertise, from battery material development to the manufacture of large-format cells, in order to enable the rapid transfer of research results into practical application.

MEFBatt: Pilotanlage zur Massenproduktion von Elektroden für Festkörperbatterien am KIT
Im BMBF geförderten Forschungsprojekt MEFBatt („Aufrüstung einer Mehrlagen-Elektrodenfertigung zur Prozessierung von Festkörper-Batterien im Pilotmaßstab“) wird am IAM-ESS des KIT der nächste Schritt zur Massenproduktion ermöglicht. Mit einer Pilotanlage soll der Fertigungsprozess von Elektroden für Festkörperbatterien zur Vorbereitung einer industriellen Massenproduktion erforscht und optimiert werden.
Zu dem Artikel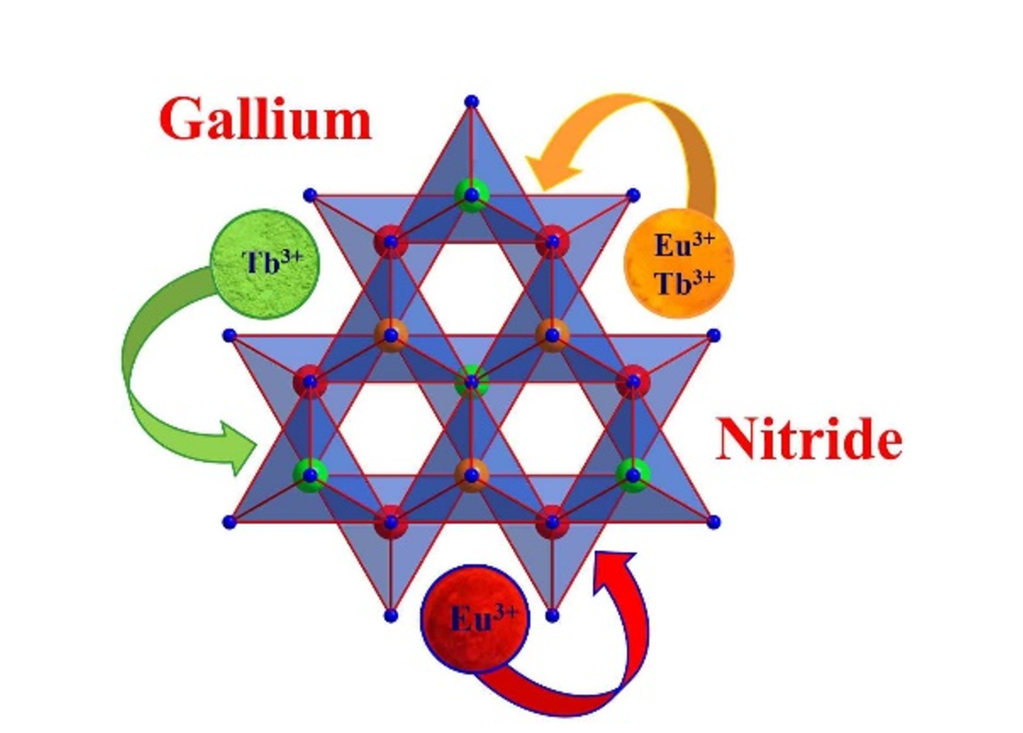
Closing the yellow gap with Eu- and Tb‑doped GaN: one luminescent host resulting in three colours
The invention of highly efficient blue-emitting diodes was a milestone for the development of today's omnipresent white LEDs being enabled by the investigations of Nakamura et al., who were awarded the Nobel prize in 2014. Here we demonstrate the doping of bulk GaN with europium, terbium and the combination of both resulting in intriguing luminescence properties, pushing the role of GaN:Eu,Tb as a chief component in future light emitting diodes. This colour tuning proves that one luminescence host can provide three colours (red, green and orange) and that even the so called “yellow gap” could be closed with a III-nitride.
Artikel aufrufen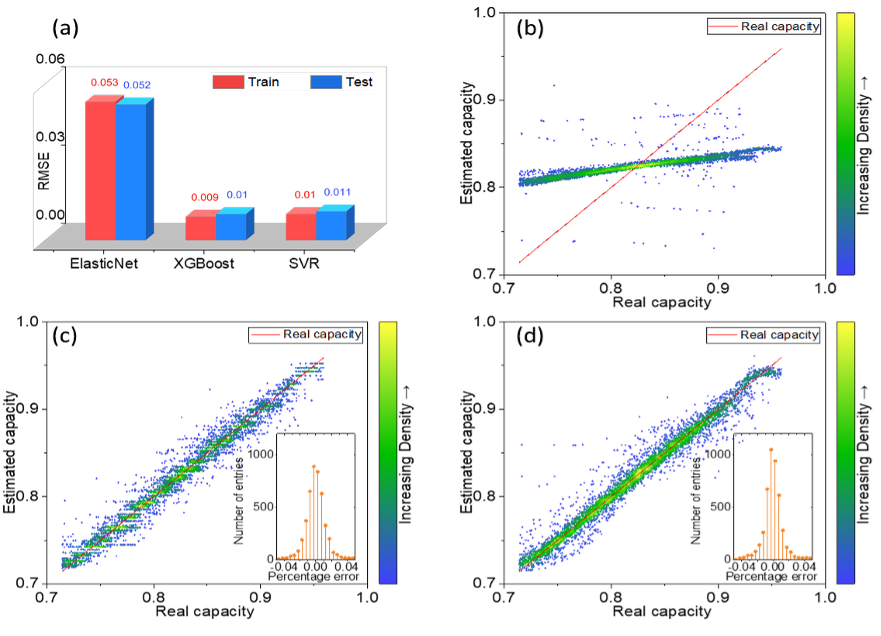
Data-driven capacity estimation of commercial lithium-ion batteries from voltage relaxation
(Article was selected as one of Editors' Highlights in section Energy:
https://www.nature.com/collections/dmmhtcypsc)
Accurate capacity estimation is crucial for the reliable and safe operation of lithium-ion Batteries. Researchers from Tongji University and KIT in cooperation with other institutes have investigated the possibility to use relaxation voltage curves of commercial cells to estimate the capacity. This way no additional cycling information and no complete charge/discharge cycle is required to determine the residual capacity. Statistical features derived from relaxation curves were compared with three different models and then used to estimate residual capacity of the cells.
Zu dem Artikel