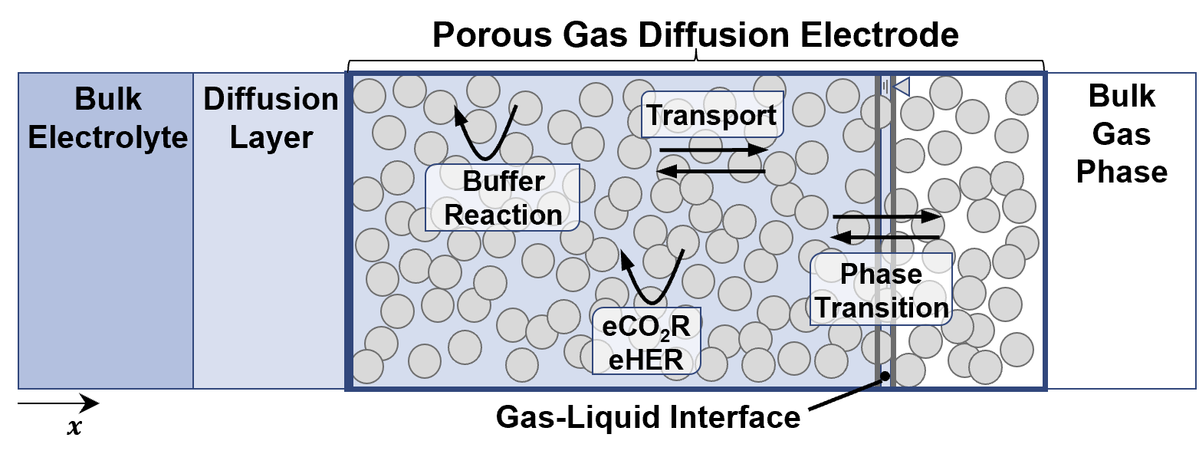
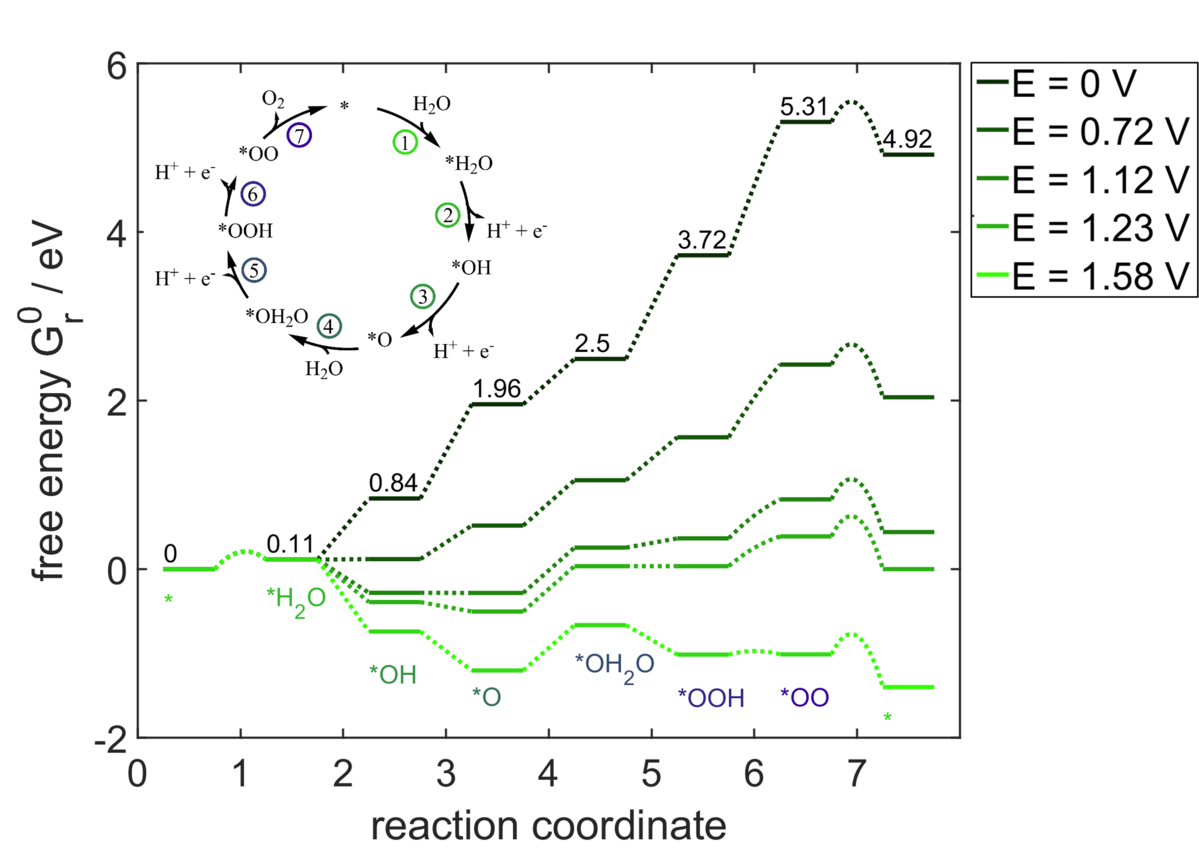
Electrocatalysis
Our research focuses on investigating electrochemical reactions at solid-liquid interfaces on electrodes and single cells, with a particular emphasis on technologies relevant to the energy transition. Specifically, we are currently studying the CO2 reduction reaction, oxygen evolution reaction in PEM water electrolysis, oxygen reduction reaction in the electrochemical H2O2 synthesis, electrolysis of biomass, and the electrosynthesis of organic chemicals. We analyze the interplay between electrode kinetics, mass transport, material properties, and product formation during electrochemical reactions. By combining physicochemical models and experimental investigations in the laboratory, we aim to identify and analyze electrocatalytic processes to gain fundamental and technically relevant insights into the interaction of these processes and their operating limits, which are crucial for the development of future-oriented technologies.
To achieve this goal, our laboratory utilizes online diagnostic and dynamic experimental methods. With our dynamic analysis approach, we can break down complex electrochemical processes into individual subprocesses by differentiating their time constants and analyzing them separately. This approach enables us to identify limiting processes and relevant parameters more precisely than conventional methods. We further support this with model-based analysis and simulations.
Our laboratory employs various experimental techniques, such as cyclic voltammetry (CV), rotating disk electrode (RDE) experiments, electrochemical impedance spectroscopy (EIS), operando electrochemical mass spectrometry (OEMS), gas or liquid chromatography coupled with mass spectrometry (HS-GC-MS, HPLC-MS), surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS), and UV-Vis spectroscopy.
Our model-based approach allows us to systematically analyze electrochemical processes from microkinetic to macrokinetic scales, including complex mass transport. Experimentally validated reaction kinetics and thermodynamic energy values serve as the basis for our physicochemical models and simulations. We apply rigorous mathematical optimization strategies to systematically improve processes or perform scenario-based analyses to determine operating parameters and process conditions.
Open positions and bachelor and master theses in our working group can be found here.
Contact: Dr. Philipp Röse (Group Leader), Swantje Pauer (Substitute)
| Topic | Contact |
|---|---|
| Carbon dioxide reduction reaction (CO2RR) on copper-based electrocatalysts (Helmholtz-Program MTET; Topic 3, Subtopic 2: Power-based Fuels and Chemicals) | |
| Dynamic kinetic multi-scale analysis of IrO2 electrodes for oxygen evolution reaction (OER) in PEM-electrolysis under technical-realistic operation conditions (H2Giga) | M.Sc. Gözde Kardes |
| Electrolysis of Biomass (ELOBIO; EIC Pathfinder Challenge: Novel Routes to Green Hydrogen Production; EIC Green Hydrogen Portfolio) | |
| Electrification of Technical Organic Syntheses (ETOS; BMFTR-Cluster4Future-Initiative) | Prof. Dr.-Ing. Ulrike Krewer, Dr. Rozalia Orghici, Dr. Philipp Röse |
| Digital Electrolyzer Design for Technical Organic Synthesis (DigiE-tos; BMFTR-Cluster4Future-Initiative) | , |
| Development and Analysis of a Large-Scale Electrolyzer for Long-Term Operation (EAGLE; BMFTR-Cluster4Future ETOS) | Dr.-Ing. Michael Küttinger |
| Laplace Pressure Controlled Gas Diffusion Electrodes for Organic Electrosynthesis (GDE4OES; BMFTR-Cluster4Future ETOS) | |
| Shared Pool of Electrode Materials and Electrolysis Cells (Pool Mazel; BMFTR-Cluster4Future ETOS) | Dr. Philipp Röse |
| ELN for electrochemical reactions (ELN Elch; BMFTR-Cluster4Future ETOS) | M.Sc. Elisabeth Oldenburg, Dr. Philipp Röse |
| LabIMotion Catalysis (KIT ExU Measure RDM) | M.Sc. Elisabeth Oldenburg, Dr. Philipp Röse |
| Synthesis and Characterization of High-Performance Anode Catalysts for use in PEM Electrolysis Cells under Realistic Conditions (MWK) | |
| Bridging Concepts in Thermo- and Electro-Hydrogen Peroxide Catalysis, Subproject 5: Microkinetic Analysis and operando FTIR spectroscopy of catalysts for e-HP (DFG Research Group FOR 5715, HyPerCat) | M.Sc. Elisabeth Oldenburg |
| Bridging Concepts in Thermo- and Electro-Hydrogen Peroxide Catalysis, Subproject 4: Multiscale Analysis of reaction-transport interactions for e-HP (DFG Research Group FOR 5715, HyPerCat) | M.Sc. Marlon Ritz |
| Topic | Contact |
|---|---|
| Multi-scale analysis of complex three-phase systems: O2 and CO2 reduction at silver-based gas-diffusion electrodes in aqueous electrolyte (DFG Research Unit 2397) | M.Sc. Inga Dorner |
| Structure-performance relationships of Ir-Ru electrodes for oxygen evolution reaction (OER) during dynamic operation (DFG Priority Programm SPP2080) | Dr.-Ing. Janis Geppert |
| Title | Type | Supervisor | Status |
|---|---|---|---|
| Einfluss von Wasserverunreinigungen auf die PEMWasserelektrolyse | Masterarbeit | Mareike Sonder, M.Sc. |
zu vergeben |
| Influence of Water Impurities on PEM Water Electrolysis Performance | Masterarbeit | open |
|
| Investigation of novel catalysts for PEM water electrolysis | Bachelor-/ Masterarbeit | open |
| Title | Type | Supervisor | Person in Charge |
|---|---|---|---|
| Electrochemical Characterization and Structural Analysis of IrCeO2-based CCMs for PEM Water Electrolysis | Masterarbeit | Beril Tüysüzoglu |
|
| Influence of the ZrO₂ Support on the OER Kinetics of Supported Iridium Catalysts | Bachelorarbeit | Tobias Zeeb |